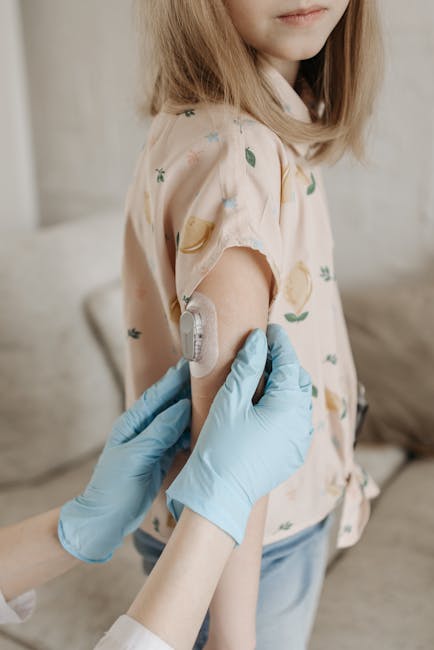
A Class I recall has been issued for certain FreeStyle Libre 3 sensors due to their potential to cause serious health consequences or death, with reports of 7 associated fatalities.
The recall indicates that the use of these continuous glucose monitoring systems poses a significant risk to users, prompting regulatory action to mitigate potential harm. A Class I recall is the most serious type of recall, reserved for situations where the use of a device could lead to severe health consequences or death.
The recall of FreeStyle Libre 3 sensors is expected to have significant implications for users who rely on these devices to manage their glucose levels. As the situation unfolds, users are advised to take immediate action to check their CGM and follow the instructions provided by the manufacturer to minimize potential risks.
















Leave a Reply